
How clinical trials shape effective skincare choices
TL;DR:
- Clinical trials validate the efficacy and safety of skincare products, enabling credible claims and building consumer trust.
- However, the quality of evidence varies; consumers should scrutinize study design, independence, and published data before relying on claims.
Walk into any pharmacy or browse a premium skincare boutique and you will encounter a wall of products boasting “clinically proven” results. But here is the reality: not all clinical claims are created equal, and the gap between a rigorous randomised controlled trial and a 12-person perception survey is enormous. Understanding how clinical trials actually work in skincare gives you a decisive advantage, allowing you to cut through the noise, invest in products that genuinely deliver, and make choices grounded in real science rather than compelling marketing copy.
Table of Contents
- What are clinical trials in skincare?
- Why clinical trial evidence matters: Beyond the label
- Regulations, claims, and real-world impact
- Not all clinical trials are equal: What to watch for
- Applying clinical trial knowledge to your skincare routine
- The uncomfortable truth: Why clinical trial results aren’t the full story
- Discover evidence-based skincare with The M-ethod
- Frequently asked questions
Key Takeaways
| Point | Details |
|---|---|
| Evidence builds trust | Robust clinical trials provide confidence that skincare products deliver on their promises. |
| Not all trials are equal | Look past simple ‘clinically tested’ labels to study design and transparency before trusting claims. |
| Regulations have limits | Skincare claims are not tightly regulated; it’s vital to check that evidence supports product statements. |
| Apply findings wisely | Using clinical trial insights helps you make more effective and safer skincare decisions. |
What are clinical trials in skincare?
A clinical trial in skincare is a structured scientific study designed to assess whether a product or ingredient performs as claimed, and whether it is safe for regular use. Unlike pharmaceutical drugs, cosmetic products are not subjected to the same mandatory pre-market approval process, yet rigorous brands still commission trials to substantiate what they put on the label.
These studies can vary considerably in scale and scope. Some involve fifty participants measured over twelve weeks using objective instruments like corneometers (which measure hydration) or spectrophotometers (which assess pigmentation). Others rely on self-reported questionnaires asking participants whether their skin “feels” firmer. Only one of those approaches produces data worth your attention.
The clinical skincare science supporting a product typically addresses one or more of the following areas:
- Efficacy: Does the product measurably improve a specific skin parameter, such as wrinkle depth, hydration, or acne lesion count?
- Safety: Does regular use cause irritation, sensitisation, or adverse reactions in a tested population?
- Functional claims: Does the evidence justify language like “reduces pigmentation by 30%” or “improves barrier function within four weeks”?
As clinical testing builds trust in premium skincare by validating efficacy and safety, it also enables brands to make substantiated claims, ensuring both consumer confidence and regulatory compliance. When a respected brand invests in this process, they are essentially putting their formulation under scrutiny. That matters.
“Clinical trials in skincare primarily validate efficacy and safety of products, enabling substantiated claims like wrinkle reduction or hydration improvement, building consumer trust and ensuring regulatory compliance for functional claims.”
The distinction between cosmetics and drugs often comes down to the depth of trial evidence. A moisturiser that “hydrates skin” sits comfortably in cosmetic territory. A product that “repairs the epidermal barrier at a cellular level” ventures toward drug-like claims and will attract greater regulatory scrutiny. Understanding this spectrum helps you contextualise the evidence behind your products.
Why clinical trial evidence matters: Beyond the label
Now that we have covered what clinical trials are, let us unpack why rigorous trial evidence makes a real difference in your skincare choices.

The phrase “clinically tested” has become almost meaningless on its own. It could mean a company tested a product on ten of its own employees for one week and found no allergic reactions. That is technically a clinical test. It says almost nothing about efficacy. Truly meaningful clinical evidence in skincare is grounded in specific design features that separate reliable data from marketing spin.
Here is a quick comparison to illustrate the difference:
| Study feature | Strong trial | Weak trial |
|---|---|---|
| Participant group | Large, diverse, controlled | Small, self-selected |
| Design | Randomised controlled trial | Open-label, no placebo |
| Measurement | Objective instruments | Self-reported perception |
| Blinding | Double-blinded | Unblinded |
| Statistical significance | p < 0.05 reported | No statistics given |
| Independent review | Peer-reviewed or third-party | Internal only |
When you see “clinically proven,” the question to ask is: proven by whom, to what standard, and published where? As studies show, consumers should verify study details including independence, p-values below 0.05, and the use of objective instrumentation, rather than accepting vague “clinically tested” labels at face value.
Here is a practical framework for evaluating a trial’s credibility:
- Check the participant numbers. Studies with fewer than 30 participants rarely have the statistical power to produce reliable conclusions.
- Look for a control group. Without a placebo or comparator, you cannot tell whether improvement is due to the product or simply time and moisturisation.
- Identify the measurement tools. Objective devices produce quantifiable data. Perception surveys do not.
- Confirm independence. Studies commissioned and conducted exclusively by the brand are prone to bias. Third-party or academic-affiliated trials carry more weight.
- Seek publication. Peer-reviewed journals impose external scrutiny. Results published only in marketing materials have not been challenged by independent scientists.
Pro Tip: When reviewing a product’s clinical evidence, look for the study’s “p-value” in the data. A p-value below 0.05 indicates statistical significance, meaning results are unlikely to be due to chance. This is a quick and reliable filter when assessing whether a claim is genuinely supported.
Split-face studies are particularly useful in skincare research. In these trials, one side of the face receives the active treatment and the other receives a control or vehicle, so each participant acts as their own comparator. This design removes much of the variability caused by individual differences in skin type, lifestyle, and genetics.
Regulations, claims, and real-world impact
Understanding how trials provide evidence is important, but it is also vital to see how regulations shape what brands can and cannot claim, and what that means for you.
In the United Kingdom, cosmetic products are regulated primarily under the UK Cosmetics Regulation, which focuses on safety, ingredient restrictions, and labelling. In the United States, the FDA governs cosmetics under a separate framework from pharmaceutical drugs. Crucially, the FDA does not require pre-market approval or clinical trials for cosmetics safety or efficacy, with the exception of colour additives. Companies self-substantiate their own safety data.
However, functional claims create a different obligation. If a product claims to “reduce wrinkles,” “fade dark spots,” or “stimulate collagen production,” the brand must have evidence to support that claim. Without it, they risk misbranding allegations or even reclassification as a drug by the FDA or equivalent authorities. This is precisely why credible brands invest in trials even when not legally compelled to do so.
Here is a snapshot of how key ingredients have performed in clinical settings:
| Ingredient | Skin concern | Trial outcome |
|---|---|---|
| Retinol | Fine lines, texture | Significant wrinkle reduction over 12 weeks |
| Niacinamide | Pigmentation, pores | Improved skin tone, reduced sebum production |
| Tea tree oil | Acne | Comparable efficacy to benzoyl peroxide |
| Hyaluronic acid | Hydration, plumpness | Measurable improvement in skin moisture levels |
| Azelaic acid | Rosacea, pigmentation | Clinically validated reduction in lesion count |
The regulatory gap between cosmetics and drugs does create a grey zone, particularly for “cosmeceuticals,” a term used to describe products that function somewhere between a cosmetic and a pharmaceutical. Products containing high concentrations of actives like retinoids, alpha hydroxy acids, or growth factors often sit in this space. These clinical skincare tips for achieving effective results often reference cosmeceuticals as the category where trial evidence becomes most critical.
- Retinol: Validated across decades of published research for stimulating collagen synthesis and accelerating cell turnover.
- Tea tree oil: Shown in well-controlled studies to reduce acne lesion counts with a tolerability profile comparable to conventional treatments.
- Vitamin C (ascorbic acid): Demonstrated efficacy in photoprotection and pigmentation reduction, though stability in formulation affects real-world outcomes.
It is also worth noting that cosmetics under US law are particularly scrutinised when anti-ageing or hyperpigmentation claims are made, since these border on drug-like territory and risk reclassification. Brands pursuing these claims therefore favour randomised controlled trials and split-face designs over perception-based surveys.
Not all clinical trials are equal: What to watch for
Even when a product boasts clinical results, there is more to the story. Here is what separates gold-standard studies from weak evidence.
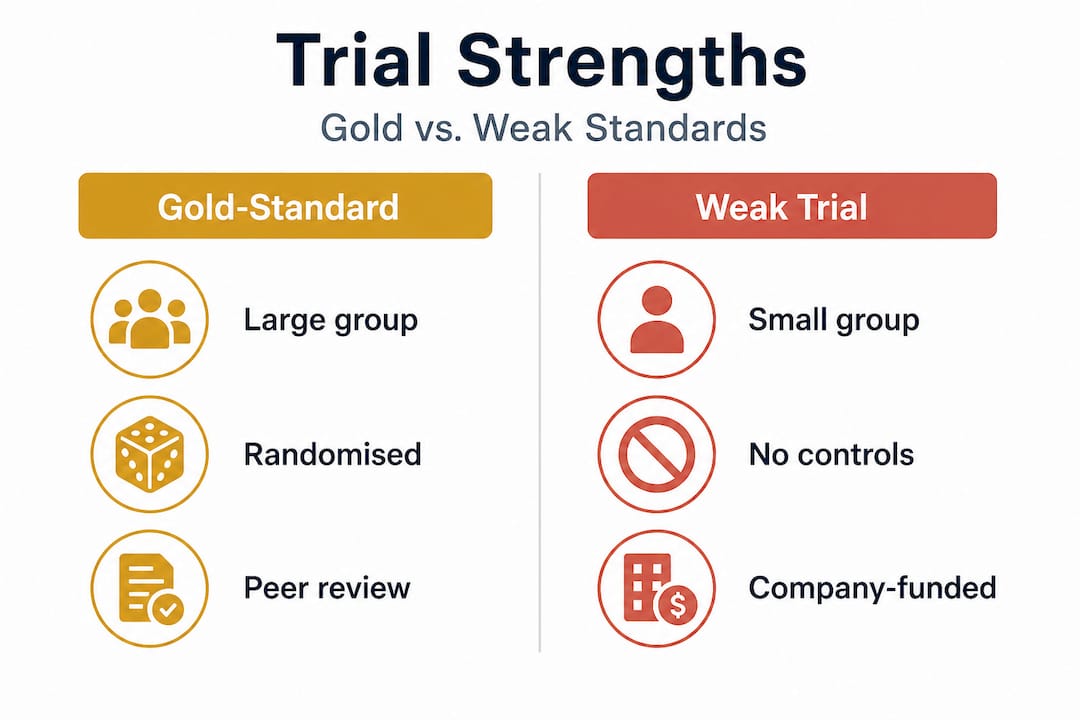
Methodological quality is not a binary judgement. It exists on a spectrum, and even well-intentioned trials can fall short. According to research on anti-ageing product testing, the key markers of rigorous design include proper blinding, appropriate controls, and participant diversity. Short studies that highlight transient effects, such as immediate hydration after a single application, are particularly prone to overstating their conclusions.
Several pitfalls appear repeatedly in lower-quality skincare trials:
- Insufficient duration. Biological changes like collagen remodelling or pigmentation correction take months, not days. Trials running for two weeks cannot reliably assess these outcomes.
- Homogeneous participants. Studies conducted exclusively on one skin tone, age group, or ethnicity limit how broadly results can be applied.
- Reliance on self-assessment. “86% of participants agreed their skin looked younger” is not the same as a statistically significant reduction in measured wrinkle depth.
- No control group. Without a group receiving a placebo or comparator, any observed improvement may simply reflect the placebo effect, seasonal hydration changes, or regression to the mean.
- Undisclosed funding. Industry-funded trials are not inherently flawed, but undisclosed conflicts of interest should raise questions.
Pro Tip: Before purchasing a product based on clinical claims, ask the brand directly whether their trial data has been published in a peer-reviewed journal. Credible brands will often direct you to a published paper or an independent summary. If none exists, treat the claim with caution.
When you are consulting a skincare expert, they can help you interpret trial data in the context of your specific skin concerns, bridging the gap between published averages and your individual skin biology. This is genuinely valuable, since even well-designed trials report average outcomes across a population, not guaranteed results for any single participant.
Applying clinical trial knowledge to your skincare routine
Equipped with these critical insights, here is how you can practically use clinical trial evidence in your daily quest for great skin.
Trial data, when read correctly, becomes a powerful filter. Consider this: a retinol pilot study involving 21 participants over 180 days showed that 76% achieved measurable improvement in fine wrinkles using the Rao-Goldman scale. A separate four-week study with 47 participants demonstrated a hydration increase of 72.5% and a reduction in dryness of 93.7%. Tea tree oil was shown to be effective for acne, with efficacy comparable to benzoyl peroxide. These are the kinds of numbers that move a claim from aspiration to evidence.
Here is a step-by-step approach for applying clinical trial knowledge to your skincare shopping:
- Identify the specific claim. Does the product promise to reduce pigmentation, improve elasticity, or clear acne? That specific claim needs specific evidence.
- Search for the supporting data. Many premium brands publish study abstracts or summaries. If the data is not available, it may not exist.
- Evaluate the study design. Apply the filters covered earlier: participant numbers, controls, objective measurement, blinding, and independence.
- Consider ingredient concentration. Clinical studies are conducted at specific concentrations. A product containing 0.025% retinol may not replicate results from a study using 0.1%.
- Assess the trial duration. Verify that results were measured over a timeframe relevant to the skin concern being addressed.
- Cross-reference with expert guidance. A virtual consultation with a clinician can contextualise published data against your skin type, lifestyle, and existing regimen.
- Monitor your own response. Even robust trials represent population averages. Track your own results with consistent photography and honest self-assessment over at least eight to twelve weeks.
The goal is not to become a statistician before buying a serum. It is to raise your baseline expectations for what counts as credible proof, so you spend wisely and achieve genuinely better outcomes.
The uncomfortable truth: Why clinical trial results aren’t the full story
Clinical trials are the most reliable tool we have for establishing whether a skincare ingredient or product works. We believe in evidence strongly, and it genuinely shapes how we curate our recommendations. But it is worth being honest about their limits.
Trials create a robust statistical baseline. They tell us that, on average, a particular percentage of participants experienced measurable improvement. What they cannot tell you is whether you will be in that percentage. Skin is extraordinarily variable. Genetics, hormonal fluctuations, stress, diet, sleep, and the interaction of other products in your routine all influence outcomes in ways no trial can fully control for.
There is also the question of industry influence on how trials are designed and communicated. Brands rarely publish studies that show their product did not perform. This publication bias means the literature skews positive. Endpoints are sometimes chosen because they are more likely to show favourable results rather than because they are the most clinically meaningful. We have seen formulations supported by technically valid trials that were designed at concentrations unlikely to appear in the finished product.
The real value of the science behind skincare results lies in combining trial evidence with expert interpretation and personal monitoring. A skilled clinician does not simply prescribe the most heavily trialled ingredient. They read your skin, contextualise the evidence, adjust for your individual biology, and iterate based on your response. That collaborative approach, grounded in science but tailored to the individual, is where lasting results genuinely come from.
Discover evidence-based skincare with The M-ethod
If you are ready to put these insights into action, The M-ethod offers a curated selection of products backed by genuine clinical evidence, selected by expert clinicians who understand the difference between robust data and clever marketing. Our PCA Clearskin acne serum is formulated with ingredients supported by well-designed efficacy studies, making it a credible choice for those managing breakouts. For those seeking clinically validated solutions across a broader range of concerns, our NEOSTRATA skincare collection represents decades of rigorous research. Pair any purchase with a virtual consultation from our team of top clinicians to ensure you are applying the right products at the right concentrations for your specific skin biology.
Frequently asked questions
What does ‘clinically proven’ really mean for skincare products?
‘Clinically proven’ means a product has been tested in a controlled setting, but study quality varies enormously. Consumers should look beyond the label for study details including independence, statistical significance, and objective instrumentation.
Are clinical trials required for skincare products?
No. The FDA does not require clinical trials for cosmetics, but functional claims such as “reduces wrinkles” must still be substantiated with credible data to avoid regulatory issues.
How can I check if a brand’s clinical trial is reliable?
Look for independent study design, such as randomised controlled trials, transparent reporting of statistical significance, and peer-reviewed publication rather than data shared only through brand marketing channels.
Do clinical trial results guarantee a skincare product will work for me?
No. Trials report average outcomes across a population, and individual results vary due to factors like skin type, genetics, hormonal status, and routine consistency. Expert guidance helps bridge this gap.
Are there specific ingredients with strong trial evidence?
Yes. Retinol and tea tree oil, for example, have demonstrated significant efficacy in well-designed studies, with retinol showing 76% improvement in fine wrinkles over 180 days and tea tree oil performing comparably to benzoyl peroxide for acne reduction.





